Les enzymes sélectionnées sont en relation avec des applications biotechnologiques comme la mise au point de médicaments sur mesure pour bloquer une pathologie ou une infection à travers l'utilisation d'inhibiteurs d'enzymes qui sont souvent des analogues de substrat (ou faux substrats) conduisant à des inhibitions compétitives vis à vis des substrats. Parmi les enzymes sujets adoptées pour cette année, on note: 1/ la cyclooxygénase (COX) et l'action de l'aspirine (acides acétyl-salicylique), 2/ la neuraminidase du virus de la grippe A H1N1 (grippe porcine) ou grippe A N5N1 (grippe aviaire) et l'action de Oseltamivir (Tameflu), 3/ la transaminase et 4/ la carboxypeptidase.
ENZYMES SUJETS:
1. Carboxypeptidase
C'est
une protéase à Zinc (appartenant aux enzymes qui
coupes les liaisons peptidiques à l'extremité C-terminale).
A l'aide de RasTop, on étudie l'enzyme complexée avec le
peptide Glycyl-tyrosine publiée sous le code 3CPA. Ci dessous
un résumé:
G
Shoham, D W Christianson, and D A Oren. 1988. X-ray crystallographic
investigation of substrate binding to carboxypeptidase A at subzero temperature.
Proc Natl Acad Sci U S A. 85, 684-688.
A high-resolution x-ray crystallographic investigation of the complex between carboxypeptidase A (CPA; peptidyl-L-amino-acid hydrolase, EC 3.4.17.1) and the slowly hydrolyzed substrate glycyl-L-tyrosine was done at -9 degrees
C. Although this enzyme-substrate complex has been the subject of earlier
crystallographic investigation, a higher resolution electron-density map
of the complex with greater occupancy of the substrate was desired. All
crystal chemistry (i.e., crystal soaking and x-ray data collection) was
performed on a diffractometer-mounted flow cell, in which the crystal
was immobilized. The x-ray data to 1.6-A resolution have yielded a well-resolved
structure in which the zinc ion of the active site is five-coordinate:
three enzyme residues (glutamate-72, histidine-69, and histidine-196)
and the carbonyl oxygen and amino terminus of glycyl-L-tyrosine complete
the coordination polyhedron of the metal. These results confirm that this
substrate may be bound in a nonproductive manner, because the hydrolytically
important zinc-bound water has been displaced and excluded from the active
site. It is likely that all dipeptide substrates of carboxypeptidase A
that carry an unprotected amino terminus are poor substrates because of
such favorable bidentate coordination to the metal ion of the active site.
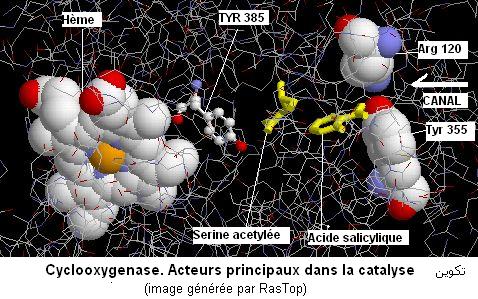
2. Cyclooxygénase
La
cyclooxygenase (l'une des enzymes particulières) intervient dans la synthèse des prostaglandines à partir
de l'acides arachidonique. L'aspirine ou acide acétylsalicylique est un anti-inflammatoire qui manifeste son effet en inhibant de façon irreversible la cyclooxygenase. A l'aide du programme RasTop, on étudie l'enzyme complexée avec un analogue de l'aspirine (l'acide 2-bromoacétoxybenzoïque) et publiée sous le code 1PTH. Ci dessous un résumé
P.J.Loll,
D.Picot, R.M.Garavito. 1995. The structural basis of aspirin activity
inferred from the crystal structure of inactivated prostaglandin H2 synthase.
Nat Struct Biol 2:637-643
Aspirin exerts its anti-inflammatory effects through selective acetylation
of serine 530 on prostaglandin H2 synthase (PGHS). Here we present the 3.4 A resolution X-ray crystal structure of PGHS isoform-1 inactivated
by the potent aspirin analogue 2-bromoacetoxy-benzoic acid. Acetylation by this analogue abolishes cyclooxygenase activity by steric blockage of the active-site channel and not through a large conformational change. We observe two rotameric states of the acetyl-serine side chain which
block the channel to different extents, a result which may explain the
dissimilar effects of aspirin on the two PGHS isoforms. We also observe
the product salicylic acid binding at a site consistent with its antagonistic
effect on aspirin activity.
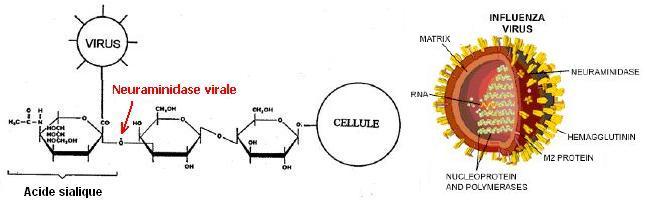
3. Neuraminidase
La
neuraminidase virale (fait partie des enzymes virales)
est une enzyme qui détache le virus préalablement fixé
sur la cellule infectée en catalysant la rupture de la liaison
qui le lie par l'acide sialique aux motifs glucidiques de la cellule.
C'est l'une des enzymes cibles pour combattre la grippe. A l'aide
du programme RasTop, on étudie l'enzyme complexée avec Oseltamivir
et publiée sous le code 2HT8. Ci dessous un résumé:
Russell,
R.J., Haire, L.F., Stevens, D.J., Collins, P.J., Lin, Y.P., Blackburn,
G.M., Hay, A.J., Gamblin, S.J., Skehel, J.J. 2006.The structure of H5N1
avian influenza neuraminidase suggests new opportunities for drug design.
Nature 2006 443:45-9.
The worldwide spread of H5N1 avian influenza has raised concerns that this virus might
acquire the ability to pass readily among humans and cause a pandemic. Two
anti-influenza drugs currently being used to treat infected patients are
oseltamivir (Tamiflu) and zanamivir (Relenza), both of which target the
neuraminidase enzyme of the virus. Reports of the emergence of drug resistance make the development of new anti-influenza molecules a priority. Neuraminidases from influenza type A viruses form two genetically distinct groups: group-1
contains the N1 neuraminidase of the H5N1 avian virus and group-2 contains
the N2 and N9 enzymes used for the structure-based design of current drugs.
Here we show by X-ray crystallography that these two groups are structurally
distinct. Group-1 neuraminidases contain a cavity adjacent to their active
sites that closes on ligand binding. Our analysis suggests that it may be
possible to exploit the size and location of the group-1 cavity to develop
new anti-influenza drugs.




4. Transaminase (Aspartate aminotransferase)
La transaminase (une des enzymes
de tranfert de groupements) est une enzyme à phosphate de pyridoxal.
Elle catalyse le transfert d'un groupe amine d'un acide aminé sur
un groupe c=o d'un cétoacide. A l'aide du programme RasTop, on
étudie l'enzyme complexée avec le glutarate et publiée
sous le code 1CZC. Ci dessous un résumé.
Shinya Oue, Akihiro Okamoto, Takato Yano and Hiroyuki Kagamiyama. 2000. Cocrystallization of a mutant aspartate aminotransferase with a C5-dicarboxylic substrate
analog: structural comparison with the enzyme-C4-dicarboxylic analog complex.
J. Biochem. 127, 337-343.
A mutant Escherichia coil aspartate aminotransferase with 17 amino acid substitutions (ATB17), previously created by directed evolution, shows increased activity for beta-branched amino acids and decreased activity
for the native substrates, aspartate and glutamate. A new mutant (ATBSN)
was generated by changing two of the 17 mutated residues back to the original
ones. ATBSN recovered the activities for aspartate and glutamate to the
level of the wild-type enzyme while maintaining the enhanced activity
of ATB17 for the other amino acid substrates. The absorption spectrum
of the bound coenzyme, pyridoxal 5`-phosphate, also returned to the original
state. ATBSN shows significantly increased affinity for substrate analogs
including succinate and glutarate, analogs of aspartate and glutamate,
respectively. Hence, we could cocrystallize ATBSN with succinate or glutarate,
and the structures show how the enzyme can bind two kinds of dicarboxylic
substrates with different chain lengths. The present results may also
provide an insight into the long-standing controversies regarding the
mode of binding of glutamate to the wild-type enzyme.
Liens utiles
- Biotechnologies, Biofertilisants et Biocontrôle (workshop national sur la Biotechnologie, Béni Mellal, Maroc, 2016)
- Biotechnologies et sécurité alimentaire (Interview)
- Pandémie de coronavirus
- تصميم الأدوية ضد الفيروسات باستهداف مراحل مضاعفة الحمض النووي و تحرير الارتباط . مقارنة بين فيروسات كورونا كوفيد 19 و الإيدز والأنفلونزا
Designing anti-virus drugs by targeting DNA duplication and release stages. Comparison between Corona Corvide 19, AIDS and influenza
- corona-covid-19, influenza, aids




ENSEIGNEMENT DE MASTER
- - Cours du Master Biotechnologies et Amélioration des Plantes, Marrakech
- Travaux pratiques Master Biotechnologies et Amélioration des Plantes, Marrakech